Sodiums atomic radius

In this case the trend increases across a period, from left to right, whereas it decreases down a group (same as with electronegativity). More accurately, ionization energy represents the amount of energy required for the removal of one or more electrons from an atom. Hence, Li will display greater electronegativity than Na.Ionization energy is often quoted as the opposite of electronegativity.
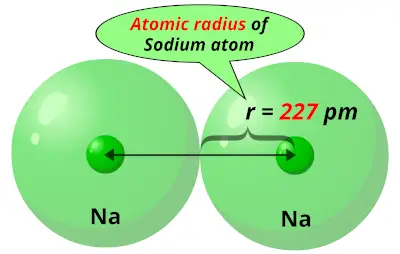
As previously mentioned, Li has a smaller atomic radius and therefore its nucleus is closer to its valence shell (and hence valence electrons) so it can exert a greater pulling force towards the attracted electrons compared to the nucleus of Na (where the valence shell is further away from the nucleus so the attractive force is less). For electronegativity, the trend that is observed is that it increases across a period, from left to right, and that it decreases as we go down a group. Therefore, overall, Na has a greater atomic radius than Li.Electronegativity is the chemical property/ability of an atom to attract electrons towards it, due to its positively charged nucleus, and form bonds. This means that in the Na atom, the electrons are further away from the nucleus and therefore less attracted by it, resulting in the atom occupying more space and therefore its atomic size being greater. Respectively for the elements of Li and Na, Li is positioned in the 2nd period and has its valence electrons in the 2nd energy levels whereas Na belongs to the 3rd period and hence has its valence electrons placed in the 3rd energy level. In the periodic table, across a period, from left to right, the atomic radius decreases whereas down a group it increases. More specifically, it refers to the half-distance between the two nuclei of two adjacent atoms of the same element. The Atomic radius of an atom represents its atomic size.
